  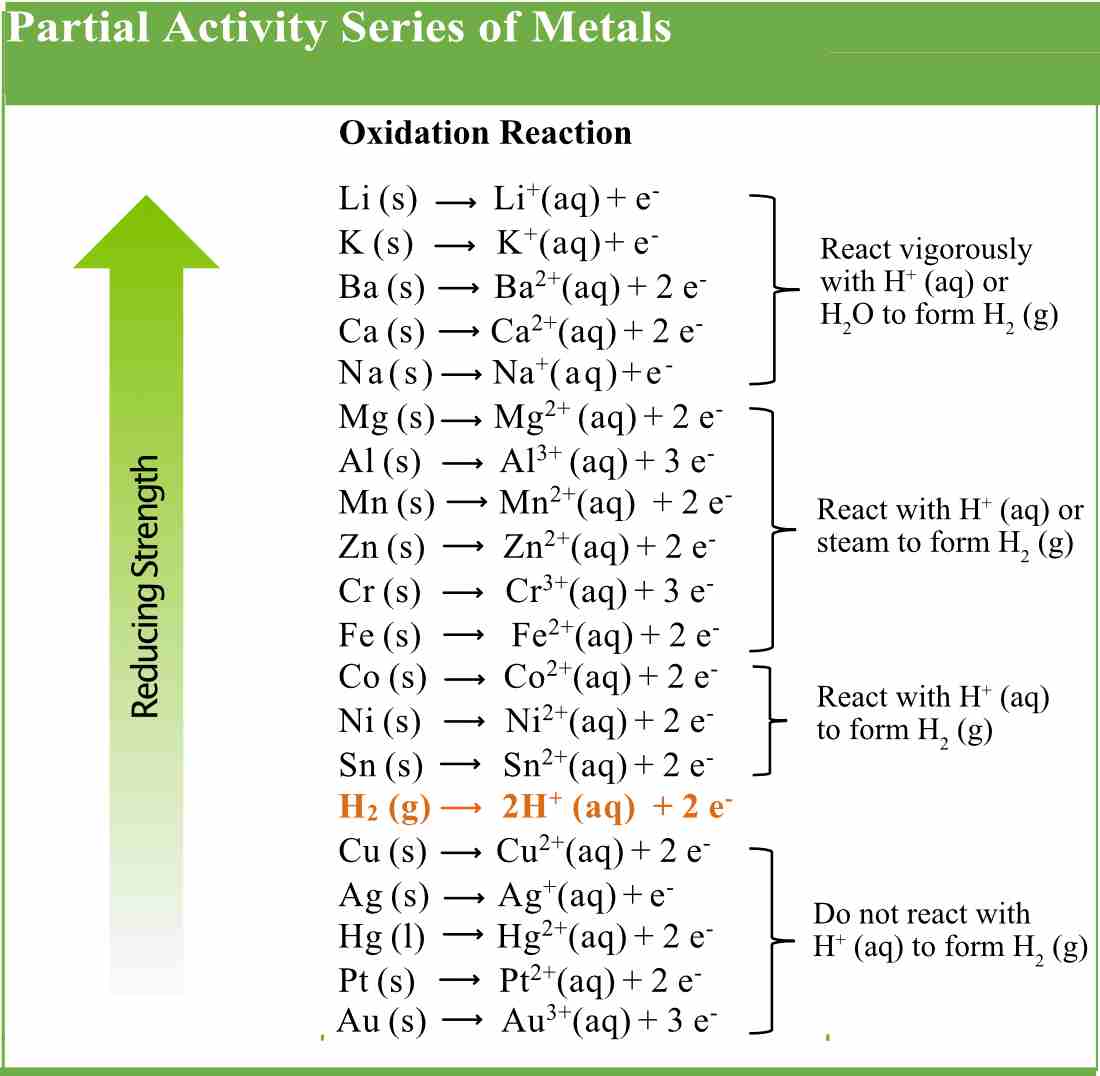
Type of AdmissionICD 10 Diagnostic GroupLegal Status Year 2006 - 2022. Students should also explain any discrepancies between their results and the publish information. Last Updated Tables HRA09 Child and Adolescent In-Patient Admissions. Jun 26 Sale +7 Colors 9 Sizes Adjustable Height Rectangular Activity Table by Norwood Commercial Furniture From 203.55 225. Also, the published version of the activity series may be compared to the results. 7 Piece Rectangular Activity Table & 21.25' H Chair Set by Flash Furniture From 171.88 1,055.00 ( 259) Fast Delivery FREE Shipping Get it by Mon. The following table describes attributes in the service data object. Relationship Type Object or Service Handled by. At the bottom are the least reactive metals. Consult the metal activity series table (page 96). Consider the two pairs of reactants listed below. The following table describes how this service data object is related to other service data objects or business object services. At the top of the list are the highly reactive metals that lose electrons during a chemical reaction to form ions. Write the balanced molecular equation, This problem has been solved Youll get a detailed solution from a subject matter expert that helps you learn core concepts. In a displacement reaction, more reactive metals displace less. Once this results have been compiled as a class, discuss how the activity aligns with the metals The instances of an appointment series that have been deleted. It will react more vigorously with chemicals It will more easily corrode This problem has been solved Youll get a detailed solution from a subject matter expert that helps you learn core concepts. The reactivity series is a list of metals arranged in decreasing order of their reactivity. They should be able to justify their decision. The students will be instructed that they are to determine the activity of each of the metals and rank them according to their results. Updated on FebruThe reactivity series is a list of metals ranked in order of decreasing reactivity, which is usually determined by the ability to displace hydrogen gas from water and acid solutions. The materials needed for this lab include 4 metal samples- such as magnesium, iron, copper, zinc, and tin-spot plates/test tubes, 0.1M HCl or 1.0M HCl, and a container for disposal of waste products. This will lead to the objective of this lab to determine the activity of four mystery metals. As students discuss that reasons for the production of hydrogen, ask whether any metal combined with HCl will produce this reaction. A possible anticipatory set for this activity would be the demonstration of the hydrogen "pop" test. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
